|
9/5/2023 0 Comments Viscosity units cst to cp 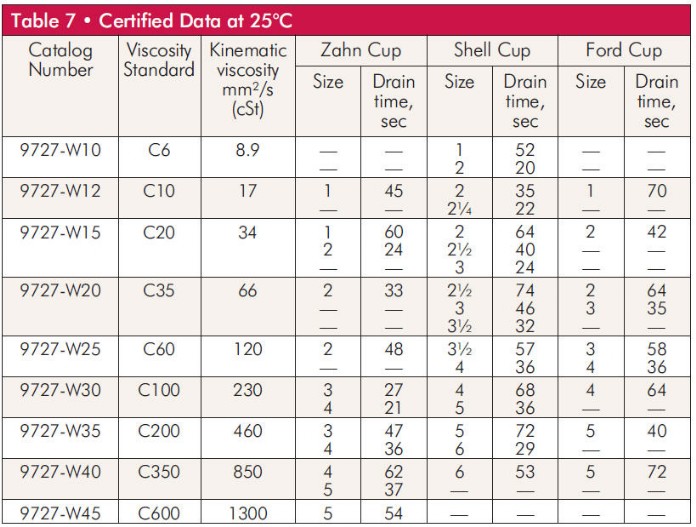
A typical plot of B o is illustrated in Figure 3.10. It relates volume of 1.0 STB of crude oil at stock tank conditions, p ST and T ST, to its volume at reservoir conditions, p r and T r. The Oil Formation Volume Factor, B o, is comparable to the water formation volume factor. This is done by placing an assumed pressure into Equation 3.42 and using Equation 3.41 to calculate the pressure associated with that assumed R so value (not the bubble-point pressure as explicitly written in the equation). When laboratory derived R so data are unavailable, then Equation 3.41 and Equation 3.42 can be used to estimate the solution gas-oil ratio. When this is done, all volumes need to be corrected back to the reference volumes of STB and SCF to get to the appropriate R so curve. The R so values are calculated by summing the appropriate gas volumes obtained during the differential test and dividing by the final oil volume. The laboratory procedure for determining the solution gas-oil ratio was discussed in terms of the differential liberation test. The volume of free gas liberated from the original stock tank barrel can be calculated as (R so i – R so) in SCF/STB or MSCF/STB. This is the behavior observed in Figure 3.09. As the gas comes out of solution and evolves into Free Gas, the volume of gas remaining in solution, R so, must decrease. 
Once the reservoir pressure reaches the bubble-point pressure, p b, gas begins to come out of solution. During this time period, the volume of gas in solution in the crude oil remains constant at the initial value of R so i. If this reservoir were to undergo pressure depletion from oil production, then the average reservoir pressure would decline over time, and the pressure would eventually reach the bubble-point pressure. The initial reservoir pressure, pi, is greater than the bubble-point pressure. In this figure, we can see that the reservoir is an undersaturated oil reservoir. When measured data are not available, Standing’s correlation can be used to estimate the bubble-point pressure: For all measurements made up to and including the bubble-point pressure, the constant composition expansion test and the differential liberation test give identical results. The constant composition expansion test is similar to the differential liberation test, however, the evolved gas is not expelled from the PVT cell during the test. Other PVT tests, such as the Constant Composition Expansion Test, can be used to determine the bubble-point pressure of the crude oil. The laboratory method for calculating the bubble-point pressure, p b, of a crude oil was discussed earlier in the context of the differential liberation test. MW o is the molecular weight of the crude oil, lb/lb-moleīubble-Point Pressure of the Crude Oil, p bĪs already discussed, the bubble-point pressure is the pressure that first bubble of gas evolves from an undersaturated crude oil during pressure reduction.It is the properties of the separated phases that we are most interested in, as these are more representative of the processes occurring in the reservoir. In addition, due to the density differences between oil and gas phases, gravity will also act to separate the two phases. Consequently, as gas evolves from the oil, this difference in the viscosity allows the gas to move faster than the oil and to separate from the source oil from which it evolved. What is the differential liberation test trying to model? In the reservoir, as gas comes out of solution, it typically has a much lower viscosity than the oil phase. The pressure, liquid volume, and gas volume are then used in the calculation of the appropriate properties for black oils. This process is then repeated until the desired final pressure is reached (Step 8). The gas is then expelled from the piston under isobaric (constant pressure) conditions by reducing the piston volume and allowing the gas to escape through a valve in the system (Step 5). At this point, the pressure and the oil and gas volumes in the cell are measured. Up until the bubble-point pressure is reached, all measurements have been single-phase (liquid hydrocarbon) measurements.Īfter the bubble-point pressure has been reached, the volume is increased further until a significant volume of free gas has developed (Step 4). This pressure is the bubble-point pressure of the crude oil. This process is continued for several pressure steps until the first bubble of gas (red) is observed through a window in the cell (Step 3). At Step 2, the pressure in the cell will be less that the original pressure due to the expansion of the crude oil. The volume of the cell is then increased by extending the piston outward (Step 2), and the pressure and volume are recorded. In a differential liberation test, a crude oil sample (green) is introduced into the cell at the initial reservoir pressure and temperature (Step 1 in Figure 3.07). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
